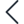If you’ve undertaken an external audit, you know the difference between being ready and not being ready.
The key challenge is to convert the effort that it takes to prepare for an audit into a corresponding benefit that will be sustained over time. Generally, though, there is limited time to prepare and consequently, the focus is on getting the evidence needed to demonstrate compliance with the standards rather than looking at the long-term value of the process to the organisation. Accreditation might be achieved, which is significant, but the potential of other benefits will quickly evaporate.
In an ideal world, the preparation would involve an in-depth review of systems and processes to ensure they are supporting safe, efficient, and effective practice. And instead of being undertaken as a once-off process every three years, the process to support ongoing review and evaluation would run deep into the organisation’s systems for managing safety, quality, and risk.
Getting ready the first time round
Standards are invaluable in providing a checklist for measuring the strength of an organisation’s governance framework. At a minimum, they detail the points where processes need to be documented (policies, procedures, protocols); where training is required (mandatory training), where checks need to be put in place to ensure practice complies with requirements (internal audits), and where evaluation to measure effectiveness would be expected (continuous improvement).
A suggestion of where to start for the initial review is to use the evidence guides that accompany the standards to identify what needs to be placed in terms of:
- Documentation requirements: policy, procedure, protocol, template, form
- Staff training requirements: work health and safety, occupation-specific
- Points where checks, monitoring, review, and evaluation are to occur
- Points where continuous improvement needs to be demonstrated
- Other requirements of the standards
Using these lists to identify what’s in place and what’s missing will make the self-assessment/gap analysis fly and will enable you to be realistic about the time and resources needed to get accreditation ready. From here, it’s all dependent on a realistic quality action plan backed up with adequate resources to support the implementation of the plan.
Getting ready the second time round
Preparation for an accreditation audit the second time round shouldn’t be about starting all over again. The first audit will have provided an understanding of what needs to be in place to meet the requirements of the standards and what that looks like for your organisation which, through the audit process, would have been validated by the auditor.
To sustain these benefits, the quality management system needs to be set up so that it can ‘pull’ the same evidence but for the timeframe of the next audit.
This was the challenge we set out to achieve with the LogiqcQMS. With one click, the platform can generate a report for the auditor detailing evidence of relevant documents, audits, improvements, training, licensing checks, and any other applicable process managed by the platform. The report can be generated for a specified timeframe (eg, last 12, 24, 36, or 48 months) and can be generated for over 40 health and related industry standards. Ideal for spot audits!
When your quality management system has this level of functionality, the focus of preparing for an audit can shift from compliance to continuous improvement, making it a far more valuable experience for the organisation.
Interested in seeing if LogiqcQMS could be the right choice for your organisation? Get in touch to arrange a free demonstration.